Introduction: Cryopreservation of peripheral blood stem cell (PBSC) or bone marrow (BM) grafts is rarely performed, thus information about the effect of cryopreservation on graft characteristics and HCT outcomes is limited. Given the global pandemic leading to changing practices for donor graft collection and increasing utilization of cryopreservation, we evaluated the outcomes of HCT recipients who received either fresh or cryopreserved allogeneic BM or PBSC grafts. The primary endpoint was engraftment. Secondary endpoints were incidence of acute graft-vs.-host disease (aGVHD), relapse, transplant related mortality (TRM), disease free survival (DFS), and overall survival (OS).
Methods: We included 7397 patients transplanted between 2013 and 2018. 1883 cryopreserved graft recipients were divided into three cohorts based on graft source and donor type: matched related (MRD) PBSC (n=1,051), matched unrelated (MUD) PBSC (n=678), and matched related or unrelated bone marrow donors (n=154). All patients received conventional calcineurin-based GVHD prophylaxis strategies. Patients who received cryopreserved grafts were matched with 5514 patients who received fresh adjusting for graft type (BM vs. PB), donor source (MRD vs. 8/8 MUD), Disease Risk Index (DRI, low risk vs. intermediate risk vs. high risk), recipient age (within 5-years) and propensity score (within 1 standard deviation from pooled sample). The propensity score was based on age, Karnofsky score, diagnosis, disease risk index, HCT-comorbidity index, and conditioning intensity. The level of statistical significance for the main analyses was p<0.01 due to multiple comparisons. The reason for cryopreservation was available on a subset of URD product recipients.
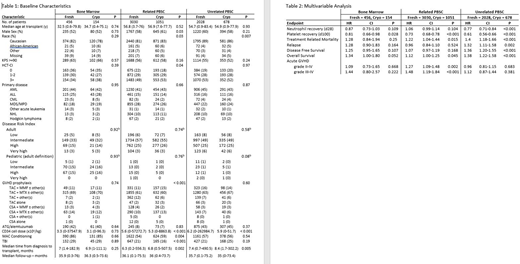
Results: Baseline characteristics for all cohorts are shown in Table 1. CD34+ doses reflect enumeration at infusion. In univariate analyses, rates of graft failure and neutrophil recovery at day 28 were similar for cryopreserved and fresh grafts for BM or related PBSC recipients but differed in cryopreserved vs. fresh MUD PBSC (5% vs 2%, p<0.001 and 87 vs 94%, p<0.001, respectively). Rates of platelet recovery within 28 days were similar in marrow cohorts, but lower with cryopreserved related (92% vs 96%, p<0.001) and MUD PBSC (87 vs 94%, p<0.001) grafts. In matched pair multivariable analyses, there were no significant differences in any outcomes with cryopreserved vs fresh BM grafts, irrespective of donor type. With related donor PBSC, cryopreservation was associated with decreased platelet recovery (HR=0.73, CI=0.68-0.78, p<0.001) and an increased risk of both grade II-IV (HR=1.27, CI=1.09-1.48, p=0.002) and grade III-IV (HR=1.48, CI=1.19-1.84, p<0.001) aGVHD, but had no impact on other outcomes. In contrast, with MUD PBSC grafts, cryopreservation was associated with delayed engraftment of neutrophils (HR=0.77, CI=0.71-0.84, p<0.001) and platelets (HR=0.61, CI=0.56-0.66, p<0.001), an increased risk of TRM (HR=1.4, CI=1.18-1.66, p<0.001) and relapse (HR=1.32, CI=1.11-1.58, p=0.002), and decreased DFS (HR=1.36, CI=1.20-1.55, p<0.001) and OS (HR=1.38, CI=1.22-1.58, p<0.001) but did not affect the incidence of aGVHD. In a subset analysis of 299 URD recipients, the most common reason for cryopreservation was delays due to patient-related events, eg., additional chemotherapy, infection. These recipients had decreased OS compared to products cryopreserved for non-patient reasons (HR=0.65, CI=0.44-0.96, p=0.029).
Conclusions: The retrospective nature of this analysis, and the fact that cryopreservation is likely to have been performed in recipients with a different (higher) risk profile compared to those receiving fresh products limits our ability to draw firm confusions. Despite these limitations, we conclude that cryopreservation in some patient populations may have a negative impact on engraftment and transplant outcomes. The decision to cryopreserve donor grafts, particularly PBSC grafts, should be carefully considered and highlights the need for further investigation of the effect of cryopreservation of allogeneic grafts where patient factors can be controlled for.
Farhadfar:CSL Behring: Research Funding; Incyte pharmaceutical: Other: Member of GVHD advisory forum. Frey:Kite Pharma: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Syntax: Consultancy, Honoraria. Devine:Magenta Therapeutics: Consultancy. Shaw:Orca Bio: Consultancy. Wingard:Shire: Other: Personal Fees - serve on data monitoring committee for clinical trial; Ansun: Other: Personal Fees - serve on data monitoring committee for clinical trial; Merck: Other: Personal Fees - serve on data monitoring committee for clinical trial; Cidara: Other: Personal Fees - serve on data monitoring committee for clinical trial; ReViral: Other: Personal Fees - serve on data monitoring committee for clinical trial.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal